|
We offer various kinds of whole home water filtration systems and offer free testing.
CLEAN DRINKING WATER THROUGHOUT THE HOME You can fill refillable water bottles for day trips and know what your drinking as well as save the planet from those plastic bottles. And, overall save money. COST SAVINGS IS THE BIGGEST FACTOR IN WHOLE HOME WATER FILTRATION SYSTEMS Cost savings. Yes, you are going to save with a whole home water filtration system. This all adds up in savings in your pockets. With less build up it will be easier to heat the water as well, saving you on energy costs. CLOTHING WILL LAST LONGER WITH A WHOLE HOME WATER FILTRATION SYSTEM Think about all the things they affect in your home. PROTECT THE ENVIRONMENT HEALTH ISSUES WITH WHOLE HOME WATER FILTRATION SYSTEMS Yes, this is a huge one as you will be making your own safe and clean drinking water. You will no longer need all those bottles of water that really aren't always safe. Save the environment from plastic bottles. You will use less soap, so there will be less soap bottles. All this plastic waste adds up and fills our landfills. You can now use reusable bottles over and over again. This is not only a cost savings but saves on plastic waste. You can fill refillable water bottles for day trips and know what your drinking as well as save the planet from those plastic bottles. Yes, you are going to save with a whole home water filtration system. With less build up it will be easier to heat the water as well, saving you on energy costs. Think about all the things they affect in your home. To learn more about clean water stop by our main site
0 Comments
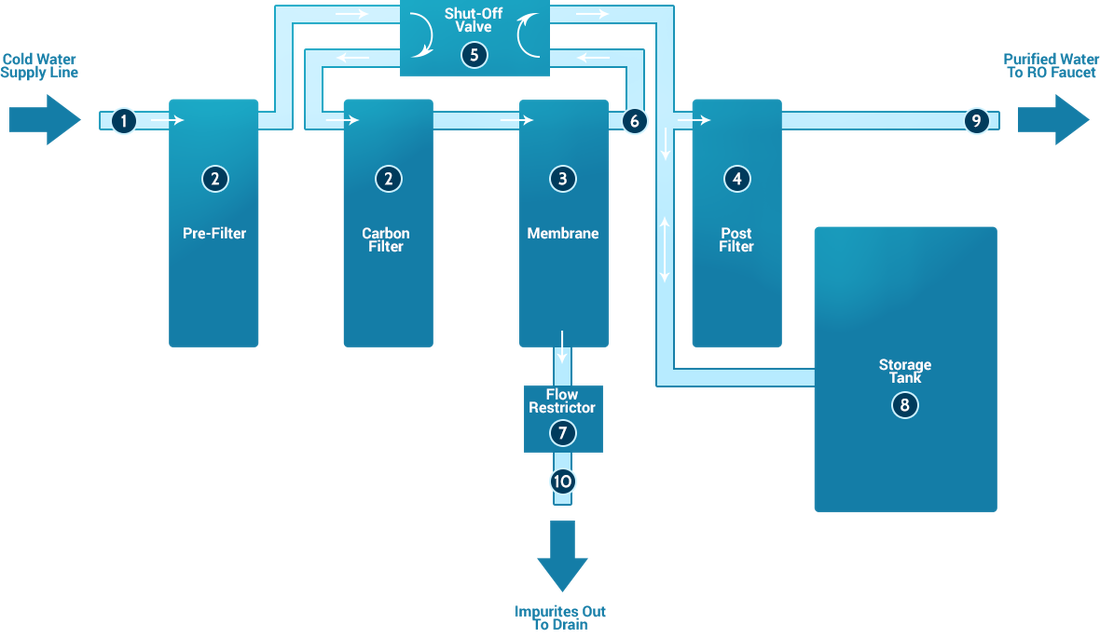
 Reverse osmosis (RO) is often a water purification technology that utilizes a semipermeable membrane to take out ions, molecules, and larger particles from waters. In reverse osmosis, an applied pressure can be used to overcome osmotic pressure, a colligative property, that is certainly driven by chemical potential differences from the solvent, a thermodynamic parameter. Reverse osmosis can remove various dissolved and suspended species from water, including bacteria, and can be used in both industrial processes and also the production of potable water. The result is that the solute is retained around the pressurized side from the membrane along with the pure solvent is able to pass for the other side. To be "selective", this membrane should never allow large molecules or ions from the pores (holes), but should allow smaller components with the solution (including solvent molecules) to feed freely. In the traditional reverse osmosis process, the solvent naturally moves from a place of low solute concentration (high water potential), via a membrane, to a space of high solute concentration (low water potential). The power for the movement on the solvent would be the reduction from the free energy from the system if your difference in solvent attention to either side of an membrane is reduced, generating osmotic pressure due to your solvent getting into the more concentrated solution. Applying an outside pressure to turnaround for the natural flow of pure solvent, thus, is reverse osmosis. The process is a lot like other membrane technology applications. However, key differences are simply between reverse osmosis and filtration. The predominant removal mechanism in membrane filtration is straining, or size exclusion, hence the process can theoretically achieve perfect efficiency no matter parameters such as the answer's pressure and concentration. Reverse osmosis also involves diffusion, making the procedure dependent on pressure, flow rate, along with other conditions. Reverse osmosis is familiar for its use in normal water purification from seawater, taking off the salt along with other effluent materials from your water molecules. HISTORY The means of osmosis through semipermeable membranes was initially observed in 1748 by Jean-Antoine Nollet. For the following two centuries, osmosis was just a phenomenon affecting the laboratory. In 1950, the University of California at Los Angeles first investigated desalination of seawater using semipermeable membranes. Researchers from both University of California at Los Angeles as well as the University of Florida successfully produced river from seawater inside mid-1950s, even so the flux was too low for being commercially viable prior to the discovery at University of California at Los Angeles by Sidney Loeb and Srinivasa Sourirajan in the National Research Council of Canada, Ottawa, of processes for making asymmetric membranes seen as a an effectively thin "skin" layer supported atop an incredibly porous and far thicker substrate region from the membrane. John Cadotte, of FilmTec Corporation, found that membranes with particularly high flux and low salt passage could possibly be made by interfacial polymerization of m-phenylenediamine and trimesoyl chloride. Cadotte's patent with this process was the main topic of litigation and has now since expired. Almost all commercial reverse osmosis membrane is now expressed by this method. By the end of 2001, about 15,200 desalination plants were functioning or inside planning stages, worldwide. In 1977 Cape Coral, Florida took over as first municipality inside United States to utilize the RO process on the large scale having an initial operating capacity of 3 million gallons each day. By 1985, due for the rapid boost in population of Cape Coral, the location had the greatest low pressure reverse osmosis plant from the world, capable of producing 15 million gallons / day (MGD) PROCESS Osmosis is often a natural process. When two solutions with assorted concentrations of any solute are separated by the semipermeable membrane, the solvent can move from low to high solute concentrations for chemical potential equilibration. Formally, reverse osmosis will be the process of forcing a solvent coming from a region of high solute concentration by using a semipermeable membrane to your region of low solute concentration by utilizing a pressure in excess in the osmotic pressure. The largest and the majority important putting on reverse osmosis would be the separation of pure water from seawater and brackish waters; seawater or brackish water is pressurized against one surface in the membrane, causing transport of salt-depleted water along the membrane and emergence of potable mineral water from the low-pressure side. The membranes used in reverse osmosis employ a dense layer inside polymer matrix—either skin of an asymmetric membrane or perhaps interfacially polymerized layer in a thin-film-composite membrane—the location where the separation occurs. In most cases, the membrane was designed to allow only water to feed through this dense layer, while preventing the passage of solutes (including salt ions). This process necessitates that a high pressure be exerted about the high concentration side with the membrane, usually 2–17 bar (30–250 psi) for fresh and brackish water, and 40–82 bar (600–1200 psi) for seawater, that has around 27 bar (390 psi) natural osmotic pressure that need to be overcome. This process is advisable known due to its use in desalination (treatment of salt and also other minerals from seawater to get water), speculate the early 1970s, it offers also been accustomed to purify freshwater for medical, industrial, and domestic applications. DRINKING WATER Around the earth, household mineral water purification systems, including a reverse osmosis step, are commonly employed for improving water for drinking and cooking. Such systems typically such as a number of steps:
In some systems, the carbon pre filter is omitted, and cellulose triacetate membrane is employed. The cellulose triacetate membrane is at risk from rotting unless protected by chlorinated water, even though the thin film composite membrane is vulnerable to breaking down ingesting chlorine. In cellulose triacetate membrane systems, a carbon postfilter is needed to clear out chlorine in the final product, water. Portable reverse osmosis water processors are offered for personal water purification in a variety of locations. To work effectively, the lake feeding to the telltale units really should be under some pressure (40 pounds per sq . in . (280 kPa) or greater may be the norm). Portable reverse osmosis water processors can be employed by people that live in rural areas without clean water, far away on the city's water pipes. Rural people filter river or ocean water themselves, as being the device is easy to work with (saline water might need special membranes). Some travelers on long boating, fishing, or island camping trips, or even in countries the location where the local water supply is polluted or substandard, use reverse osmosis water processors as well as one or more ultraviolet sterilizers. |
ArchivesCategories |

 RSS Feed
RSS Feed
